 The salt bridge can even contain a gel material containing the ionic. 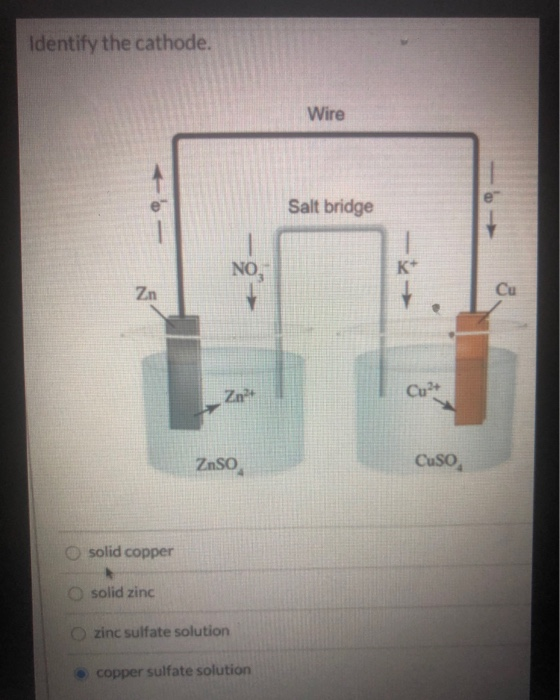
If you measure the voltage under load, it will also be affect by the nature of the salt bridge. A salt bridge is a u-shaped tube plugged at each end with cotton wool that contains an aqueous solution of an ionic salt. The nickeliron battery (NiFe battery) is a rechargeable battery having nickel (III) oxide-hydroxide positive plates and iron negative plates, with an electrolyte of potassium hydroxide. Dissolved positive and negative ions in the salt bridge can flow in order to restore charge imbalance caused by the flow of electrons from the battery. If ion transport along the salt bridge is the rate-determining step, it will affect the current (current is movement of charge per time). A salt bridge in certain batteries is placed between the anode and cathode of the battery, were electrons leave and enter the battery, respectively. The voltage and the current depend on what load you attach to the voltaic cell, and properties of the cell itself. Select from a diagram of your best battery cathode, anode, electrolytes, electron flow, salt bridge, and salt bridge ions flow and two half-reactions. A similar (but reversed) situation is found in the cathodic cell, where $\ce$ in making the salt bridge have any effects on voltage/current output of the cell? why? In order to maintain neutrality, the negatively charged ions in the salt bridge will migrate into the anodic half cell. The anions from the salt bridge diffuse into the solution on the anode side, and the cations of the salt bridge flow into the solution of the cathode side. The electrons move through the wire (and your device, which I haven't included in the diagram), leaving the unbalanced positive charge in this vessel. 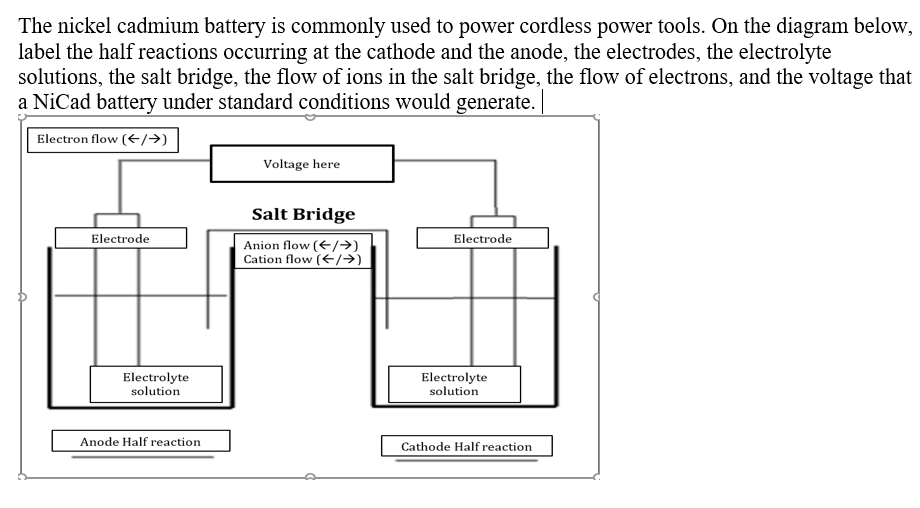
In lead storage battery, both cathode and anode are dipped in the same solution. I assume after some time cations accumulate around the anode and anions accumulate around the cathode, inhibiting further flow of electric charge. In modern batteries, salt bridges are often referred as separators. When I was stirring the water again, the current was increasing again to 30mA and was decreasing again thereafter. 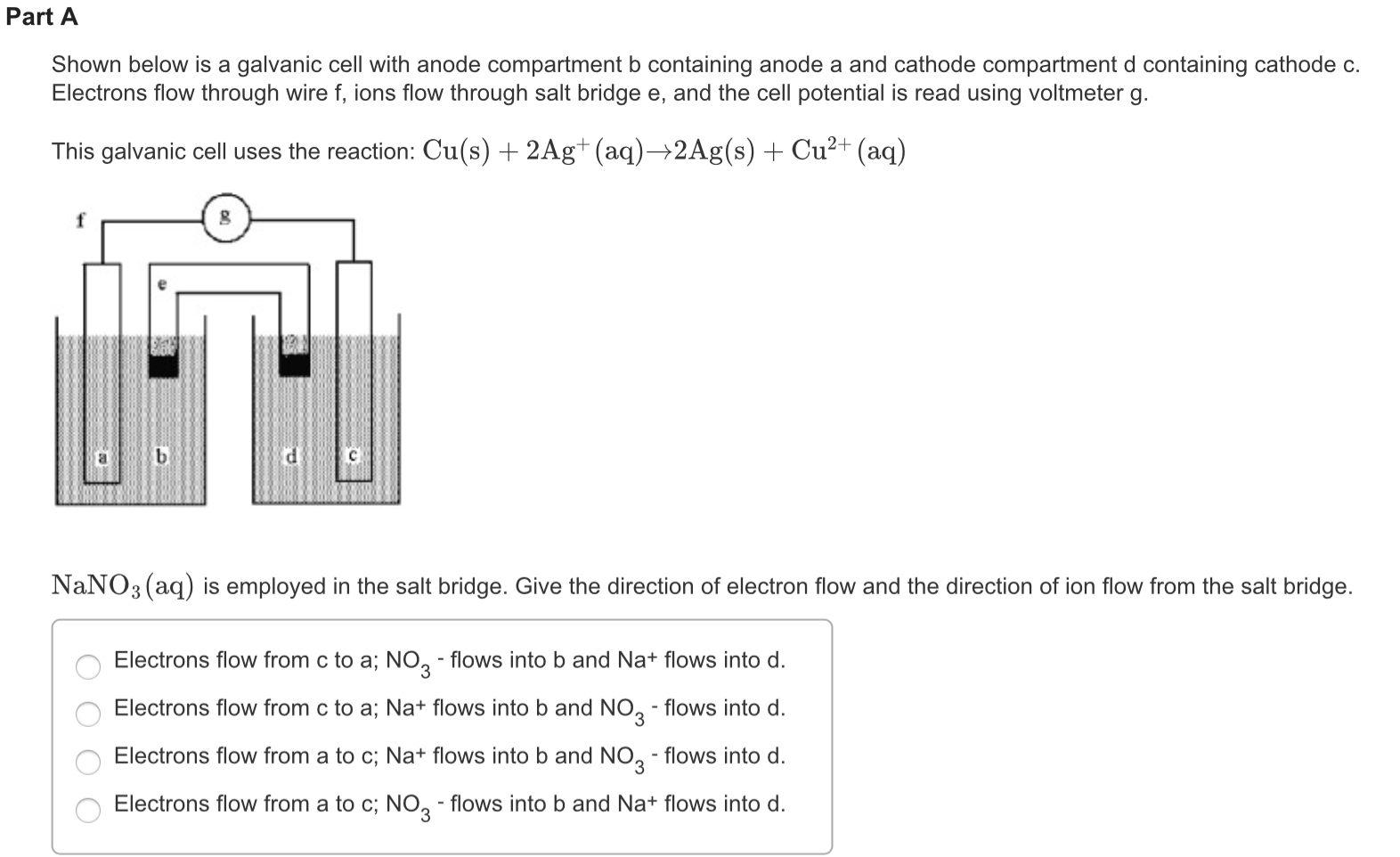
The oxidation reaction that occurs at the anode generates electrons and positively charged ions. After adding salt and stirring the water, it showed 30mA. The electrons flow from the anode to the cathode. The two half-cells are linked by a salt bridge carrying ions between them. For instance, a typical 12V leadacid battery has six galvanic cells connected in series with the anodes composed of lead and cathodes composed of lead dioxide. Which additional item is required for the construction of the voltaic cell A) Sn2+ Sn4+ + 2e. The purpose of a salt bridge is not to move electrons from the electrolyte, rather it's to maintain charge balance because the electrons are moving from one-half cell to the other. A demonstration electrochemical cell setup resembling the Daniell cell. A short Standard Reduction Potentials table is available from the HandbookMenu, but you may also click the live link to see one.There's another question related to salt bridges on this site. The anode is connected to the cathode in the other half-cell, often shown on the right side in a figure. These values are usually tabulated in handbooks. Reduction potentials of standard cells have been measured against the SHE or other standards their potentials are measured. Ideally, for every redox couple, there is a reduction potential. The electrode where oxidation occurs is called the anode. \( \newcommand\) ions are less ready to accept electrons than hydrogen ions. The salt bridge also prevents the two reacting solutions from mixing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
